Cancer
Sugar Leads To Obesity And Feeds Cancer Growth
Published
4 years agoon

As if gaining weight is not a good enough reason to reduce sugar intake, here’s another report that repeats the fact that cancer feeds off sugar. We’ve known that for a long time, but people don’t seem motivated enough by it to make any changes in their lives.
So this ties a number of healthy dietary suggestions together: cut back on the desserts, each much fewer carbohydrates (more protein would be better), and take more antioxidants. A recent article called “Glucose Deprivation Activates Feedback Loop That Kills Cancer Cells” in Medical News Today is sure to spawn new studies in the ongoing battle to treat, and beat cancer.
And although it’s not mentioned in this article, other simple things you can do to stay healthy include keeping your body pH high enough to remain alkaline, instead of acidic, and ensuring that you supply your cells with plenty of oxygen, since most diseases occur in an oxygen-depleted environment. I plan to write more about that in a future article.
Compared to normal cells, cancer cells have a prodigious appetite for glucose, the result of a shift in cell metabolism known as aerobic glycolysis or the “Warburg effect.” Researchers focusing on this effect as a possible target for cancer therapies have examined how biochemical signals present in cancer cells regulate the altered metabolic state.
UCLA research
Now, in a unique study, a UCLA research team led by Thomas Graeber, a professor of molecular and medical pharmacology, has investigated the reverse aspect: how the metabolism of glucose affects the biochemical signals present in cancer cells.
In research published in the journal Molecular Systems Biology, Graeber and his colleagues demonstrate that glucose starvation – that is, depriving cancer cells of glucose – activates a metabolic and signaling amplification loop that leads to cancer cell death as a result of the toxic accumulation of reactive oxygen species, the cell-damaging molecules, and ions targeted by antioxidants like vitamin C.
UCLA scientists
The research, which involved UCLA scientists from the Crump Institute for Molecular Imaging, the Institute for Molecular Medicine, the California NanoSystems Institute, the Jonsson Comprehensive Cancer Center, the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research, and the Department of Pathology and Laboratory Medicine, demonstrates the power of systems biology in uncovering relationships between metabolism and signaling at the network level.
“Most strikingly, our discovery that glucose withdrawal causes both cell death and increased tyrosine phosphorylation is intriguing because increased tyrosine kinase signaling is normally associated with cell growth,” said Nicholas A. Graham, a senior postdoctoral scholar in Graeber’s lab who helped design the project
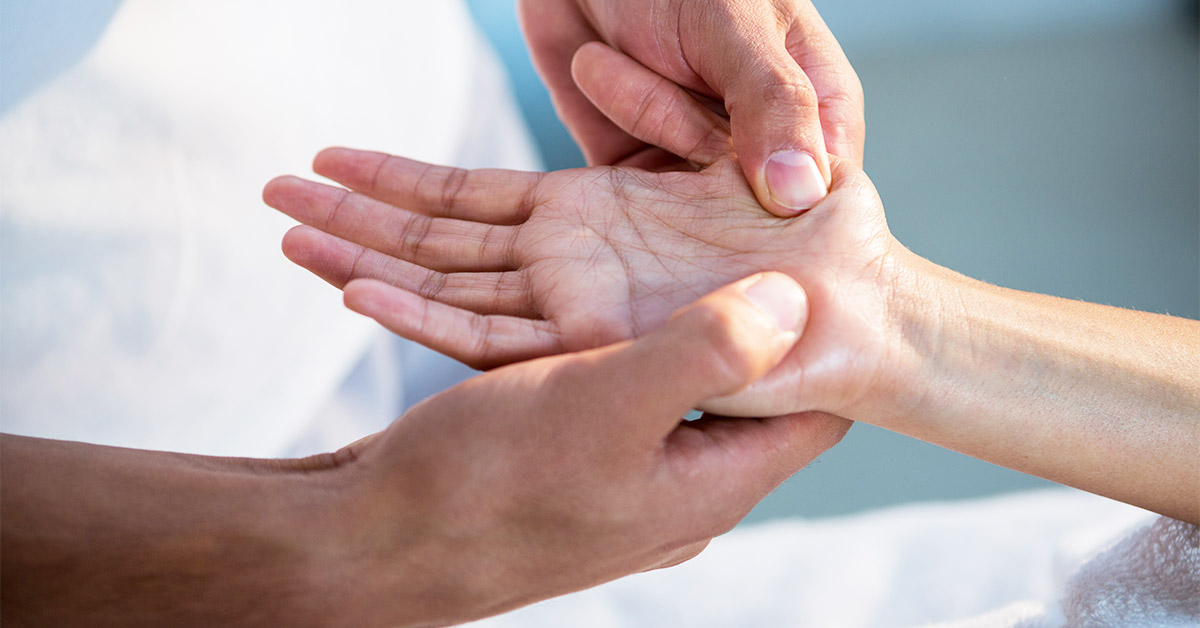
This article is liable to be thought of as “Woo Hoo” by some readers, so if you don’t believe in energy medicine, you’re likely best to stop reading now. Still here? Glad to see you. In a report called “Touch Therapy May Reduce Pain, Nausea In Cancer Patients” in Medical News Today, we learn of a new treatment that has proven beneficial in decreasing some of the symptoms of cancer.
Jin Shin Jyutsu website
As stated on the Jin Shin Jyutsu website, “Jin Shin Jyutsu is the Art of releasing tensions which are the causes for various symptoms in the body. Our bodies contain several energy pathways that feed life into all of our cells. When one or more of these paths become blocked, this damming effect may lead to discomfort or even pain. This blockage or stagnation will not only disrupt the local area but will continue and eventually disharmonize the complete path or paths of the energy flow.”
I first learned of Emotional Freedom Techniques about 10 years ago, which is the practice of using your fingertips to tap on various acupuncture points of the body (mostly on the face), to release energy blockage and promote better health. The two methods seem somewhat related, and while EFT can be used on dozens (hundreds?) of physical and emotional problems, this article only discusses cancer treatment. Please read on for more.
University of Kentucky Markey Cancer Center
A new study by the University of Kentucky Markey Cancer Center shows that patients reported significant improvement in side effects of cancer treatment following just one Jin Shin Jyutsu session. Jin Shin Jyutsu is an ancient form of touch therapy similar to acupuncture in philosophy.
Presented at the 2012 Markey Cancer Center Research Day by Jennifer Bradley, who is the Jin Shin Jyutsu integrative practitioner at Markey, the study included 159 current cancer patients. Before and after each Jin Shin Jyutsu session, Bradley asked patients to assess their symptoms of pain, stress, and nausea on a scale of 0-10, with 0 representing no symptoms.
The study found that in each session patients experienced significant improvement in the areas of pain, stress, and nausea with the first visit and in subsequent visits as well. The mean decreases experienced were three points for stress and two points for both pain and nausea.
Jin Shin Jyutsu made improvements
“I was pleased to see quantitatively the improvements that patients noted in these primary areas of discomfort,” said Bradley. “It was interesting to note that regardless of age, sex, or diagnosis, cancer patients received a statistically significant improvement in the side effects of treatment. It is encouraging to note that Jin Shin Jyutsu made improvements in these areas without adding additional unwanted effects that so often occur with medication interventions.”
Lexington Cancer Foundation

Funded by a grant from the Lexington Cancer Foundation, Jin Shin Jyutsu is considered part of an integrative treatment plan available at the UK Markey Cancer Center. Bradley offers Jin Shin Jyutsu to all cancer patients at no charge. Patients may self-refer, though half are referred by their physician or
Markey staff.

During a Jin Shin Jyutsu session, patients receive light touches on 52 specific energetic points called Safety Energy Locks as well as fingers, toes, and midpoints on the upper arm, upper calf and lower leg in predetermined orders known as “flows.” Patients remained clothed except for shoes, and all hand placements are done over clothing

A recent report titled “The Surprising Answer To Why Cancer Rate Increases With Age” published in Medical News Today is bound to stir up some controversy. While we know that cancer occurs more in the elderly, a new theory suggests why.
Young people have lots of healthy cells and young tissues that are related in such a way that they can defeat cancer, but as we age, this relationship changes, and it is thought that cells’ existing mutations tend to favor the survival of cancerous cells more. It took me a while to understand this theory, but now I can see the potential accuracy of it, and I suggest you read on for more details.
Cancers are age-related
Cancers are age-related, much more frequent in the old than in the young. A University of Colorado Cancer Center review published in the journal Oncogene argues against the conventional wisdom that the accumulation of cancer-causing mutations leads to more cancer in older people, instead positing that it is the changing features of tissue in old age that promote higher cancer rates in the elderly.
“If you look at Mick Jagger in 1960 compared to Mick Jagger today, it’s obvious that his tissue landscape has changed,” says James DeGregori, Ph.D., an investigator at the University of Colorado Cancer Center and professor of molecular biology at the University of Colorado School of Medicine. “And it’s this change, not the accumulation of cancer-causing mutations, that drives cancer rates higher as we grow older.”
For evidence, DeGregori points first to the fact that by the time we stop growing in our late teens, we’ve already accumulated a large fraction of the mutations we will have in our lifetimes. “There’s a mismatch between the mutation curve and the cancer curve,” DeGregori says, meaning that if cancer were due to reaching a tipping point of, say, five or six mutations, we should see higher cancer rates in 20-year-olds, as this is when mutation rate is highest.
DeGregori points
Second, DeGregori points out that even healthy tissues are full of oncogenic mutations. “These mutations are many times more common than the cancers associated with them,” DeGregori says. Simply, more mutations don’t equal more cancer – not across the aging population and not even in specific tissues.
DeGregori’s final two points come from evolution. As we’ve evolved from one-celled, short-lived life forms into multicellular, long-lived humans, we’ve had to develop complicated machinery to maintain our tissues and avoid disease
Trending

 digestive health4 years ago
digestive health4 years agoRelationship Between Fiber And Antioxidants

 Health4 years ago
Health4 years agoBest & Simple Core Strengthening Exercises That You Have to Do At Home

 Health4 years ago
Health4 years agoExercises To Strengthen Knees – Know Before You Go!

 digestive health4 years ago
digestive health4 years agoThe Digestive System Explained

 digestive health4 years ago
digestive health4 years agoIs Gluten-Free Food Tax Deductible?

 Fish Oil4 years ago
Fish Oil4 years agoFish Oil Has Far-Reaching Benefits

 Weight Loss4 years ago
Weight Loss4 years agoHealth Benefits Of Walking Daily

 Fish Oil4 years ago
Fish Oil4 years agoFish Oil Health Benefits








